
The year is 2048. A new economic war is raging. Rare earth has become a key element in the construction of advanced new machinery, and a major economic force around the world.
Private companies are arming the latest prototype submarines to dive into the world's depths in search of rare earth. Under the peaceful surface of the sea, a silent war is being fought with new and unstable technology.
You are part of a team of elite officers in command of a state-of-the-art submarine.
To beat the enemy, you must cooperate with your teammates. Only the team who best communicates and works together will survive the encounter...
In Captain Sonar, two teams operate opposing submarines battling for control of the ocean's depths. each teammate has his own station and responsibilities aboard the vessel. the players will have to coordinate and communicate effectively to survive.
Important: there are two different ways to play: real-time mode and turn-by-turn mode. for a first game, or if you are playing in a noisy environment, we suggest you play the turn-by-turn mode. however, the real-time mode will guarantee that you get the stress and adrenaline rush of a real submarine duel!
Components
2 sets of 12 role sheets (1 for each team, yellow and blue). Each sheet is two-sided: the light side is used for turn-by-turn mode, while the dark side is used for real-time mode.
For each team:
- 1 First Mate sheet
- 1 Engineer sheet
- 5 Captain sheets (1 for each scenario)
- 5 Radio Operator sheets (1 for each scenario)
- 2 transparent sheets for the Radio Operators
- 8 erasable marker pens
- 2 screens

Object of the Game
The first team whose submarine suffers 4 damage loses the game. The surviving team wins!
Some advanced scenarios contain additional objectives or replacement objectives that the teams must complete in order to win the game.
Setup
Game Setup
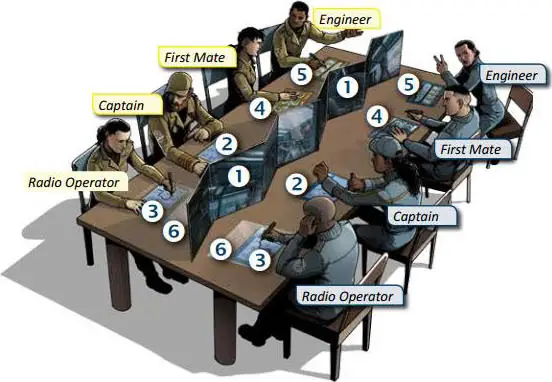
Place both screens in the center of the table end-to-end 1.
2 Choose a scenario. Each Captain 2 and Radio Operator 3 takes the corresponding sheet.
When playing in real-time mode, players use the dark side of their sheet. In turn-by-turn mode, players use the light side.
Each team sits on a different side of the central screens, with players sitting in the order shown in the diagram.
Each First Mate 4 and Engineer 5 takes a corresponding sheet.
Each Radio Operator takes a transparent sheet 6.
Finally, each player takes an erasable marker pen.
Note: Each sheet has a space for the player's name and the name of the submarine (team name).
Team Lineup
- 8 players: 2 teams of 4
- 7 players: 1 team of 4 and 1 team of 3
- 6 players: 2 teams of 3
- 5 players: 1 team of 3 and 1 team of 2
- 4 players: 2 teams of 2
- 3 players: 1 team of 2 and 1 player alone
- 2 players: 2 players alone
In a 2- or 3-player game, players must use turn-by-turn mode, as one player will need more time to manage all four roles by himself.
Role Assignments Depending On The Number Of Players
- Teams of 4: Each player plays a different role.
- Teams of 3: One player plays both the Captain and First Mate roles.
- Teams of 2: One player plays the Captain, First Mate, and Engineer roles.
The Four Different Roles Aboard
The Captain

The Captain is the leader of the team, responsible for guiding the submarine and for coordinating the team members.
At the beginning of the game, the Captain draws an "X" on the sea space of his choice to determine the starting position of his submarine. This is not shown to the enemy team.
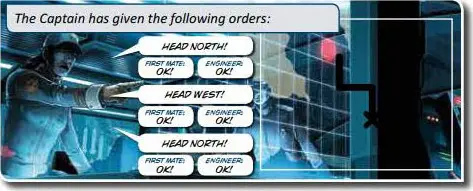
Then both Captains shout "DIVE" to signal the start of the game. Each captain announces the course of his submarine and draws the route on the map on his sheet. In turn-by-turn mode, randomly determine a team to take the first turn.
Announcing A Course
The Captain can move his submarine one space at a time in one of four directions: north, south, east, or west.
He must announce his course loudly and clearly so that all players can hear it. The announcement should be phrased as "HEAD NORTH/SOUTH/EAST/WEST".
real-time mode: Each Captain announces his course at the pace of his choice.
turn-by-turn mode: The Captains alternate announcing a course, allowing as much time as necessary for all players to complete their tasks.
Important : In both game modes, the Captain must wait for the First Mate and the Engineer to perform their tasks and announce "OK" before announcing a new course.
A submarine cannot cross or move along its own previous route (i.e. move into a space where a line is already drawn).
A submarine cannot move across an island or into a space with its own mine (see Dropping a Mine, page 6).
The Captain can erase his route only by surfacing.
Blackout!
If the Captain cannot announce a course due to islands, mines or the submarine's route, he MUST IMMEDIATELY SURFACE.
Advice:
The Captain should regularly check with his Radio Operator to get an estimate of the enemy's position.
The Captain should not announce his courses too quickly, so that his teammates have time to react and perform their tasks.
The Radio Operator

The Radio Operator is the eyes and ears of the team. He is responsible for tracking the enemy submarine by listening to the enemy Captain's announcements.
Each time the enemy Captain announces a course, the Radio Operator draws it on his transparent sheet (beginning anywhere, since he does not know the enemy's starting location).
By sliding his transparent sheet around his map, the Radio Operator will be able to estimate the enemy submarine's position, bearing in mind that the enemy cannot cross islands or its own route.
The more the Radio Operator is able to listen to the enemy Captain, the more easily he will be able to determine the enemy's route and position. He will need to regularly update his Captain with information on the possible location of the enemy.
The Radio Operator will be able to narrow the enemy's position down further with the help of drones and sonar, which will be activated by the Captain or the First Mate.

Advice:
To avoid drawing outside of the transparent sheet, try to start in the middle of the sheet at the beginning of the game.
If the route goes outside of the transparent sheet, or if you become lost, erase everything and start over.
The First Mate

The First Mate's job is to advance the gauges to bring the submarine's systems online. He alerts the Captain when the weapon, detection, and special systems are ready to activate.
Managing The Gauges
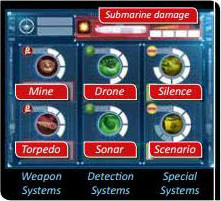
Each time the Captain announces a course, the First mate marks an empty space on the gauge of his choice.
When all spaces on a gauge are marked, the First Mate announces it loudly and clearly. For example, an announcement of "MINE READY" lets the Captain know that he can drop a mine when he needs it.
When all spaces on a gauge are full, the system must be activated before the spaces can be marked again.
Only the Captain can activate the mine, torpedo, silence, and scenario systems. The drone and sonar systems can be activated by both the Captain and the First Mate

Damage

At the top of the First Mate's sheet are four damage spaces representing the 4 damage a submarine can sustain.
Each time the submarine suffers damage, the First Mate crosses out the leftmost empty space. If all 4 spaces are crossed out, the submarine is destroyed and its team loses the game.
IMPORTANT: If you play with fewer than 8 players, a Captain might have to play the role of the First Mate as well (and maybe even of the Engineer). If this is the case, each time the Captain gives an order, he immediately marks a space on the gauge of his choice on the First Mate sheet (and a breakdown on the Engineer sheet if he is also playing that role).
The Engineer

The Engineer's job is to track the breakdowns that occur on the submarine as the Captain announces courses. Some breakdowns will neutralize the submarine's systems; others will damage the submarine.
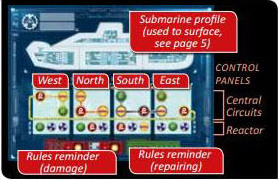
Each time the Captain announces a course, the Engineer crosses out a symbol corresponding to the announced course (north, south, east, or west), signifying a breakdown. He can cross out any symbol in the Central Circuits or in the Reactor, as long as it is in the correct control panel.

System Breakdown
If at least one symbol corresponding to a system is crossed out, that system cannot be activated.
IMPORTANT: Each symbol corresponds to two systems.
 MINE + TORPEDO
MINE + TORPEDO
 DRONE + SONAR
DRONE + SONAR
 SILENCE + SCENARIO
SILENCE + SCENARIO
Before activating a system, the First Mate must verify with the Engineer that there are no breakdowns on that system.

Radiation Breakdowns
Radiation breakdowns have no effect unless ALL radiation symbols are crossed out or they contribute to a complete area breakdown (see Complete Area Breakdown, below).
When all radiation symbols are crossed out, the submarine suffers 1 damage.
The Engineer announces "DAMAGE", then erases ALL breakdowns from the submarine.
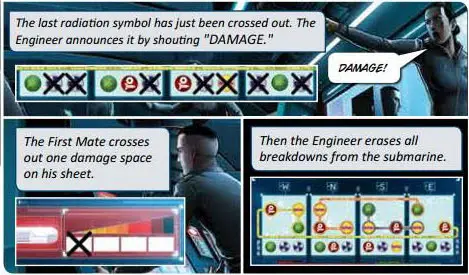
Complete Area Breakdown
When all symbols in a single control panel have been crossed out (in both the Central Circuits and the Reactor), the submarine suffers 1 damage. The Engineer announces "DAMAGE", then erases ALL breakdowns from the submarine.

Repairing
Self-repairing In The Central Circuits
Symbols in the Central Circuits are linked in groups of four by an orange, yellow, or gray line called a circuit.
When all four symbols on a circuit are crossed out, they repair themselves; the Engineer erases those breakdowns.
Breakdowns in the Central Circuits can also be repaired by surfacing.

Repairing The Reactor
Breakdowns in the Reactor can be repaired in two ways:
When all radiation symbols have been crossed out, the submarine suffers 1 damage. The Engineer announces "DAMAGE", then erases ALL breakdowns from the submarine.
The best way to repair the Reactor is by surfacing.

Surfacing
Surfacing is an efficient way to repair the submarine and the only way to reset the route. Doing so will leave the submarine vulnerable for a short time, so surfacing should be undertaken with the utmost caution.
To surface, the Captain raises his fist and announces "SURFACE".
Then the Captain announces the submarine's current sector to the enemy team.

The rules for surfacing vary depending on whether the game is being played in turn-by-turn or real-time mode:
Turn-by-turn
Instead of announcing a course, the Captain spends his turn announcing that he is surfacing, as described above. Then the Engineer erases ALL breakdowns from the submarine.
The enemy team takes three turns in a row. If the team surfaces during those turns, any remaining turns are lost. Then the game resumes as normal.
Real-time
As soon as the submarine's sector is announced, repairs can begin:
-
The Engineer begins securing the ship by drawing a line around one of the four sections of the submarine on his sheet, making sure to stay within the white outline. Then he writes his initials inside that section.
-
He passes the sheet to a teammate, who does the same with another section of the submarine and continues passing it on until all sections are secured (including all teammates' initials). If there are fewer than four players on a team, some players will need to secure multiple sections.

-
The Engineer takes the sheet and shows it to the enemy Engineer to confirm that no lines were drawn outside the white outline. If this is not the case, the team must start over.
-
If the enemy Engineer confirms that all lines are drawn correctly, the Engineer erases all lines around submarine sections, all initials, and ALL breakdowns from the submarine.
-
When this is complete, the Engineer announces "READY TO DIVE". Then the Captain announces "DIVE" and play resumes as normal.
Important: While a team is surfaced and repairing, the enemy team continues playing. Teammates on a surfaced submarine can use their marker pens only to secure the submarine.
In the meantime, the Captain resets his route by erasing the route on his sheet, keeping only the submarine's current position and the positions of his mines.

Advice
Once again, communication is the key to success.
The Engineer MUST inform the Captain of breakdowns. The Captain will be able to decide to surface when he thinks he is safe and far from the enemy (though only the Radio Operator can guarantee that!). This will allow for uninterrupted repairs and preparation for a new route.
Surfacing when the enemy is nearby is risky, and can leave a submarine vulnerable to a killing blow!
Surfacing can also be used to cleverly lure the enemy into a minefield, which can be activated as soon as the submarine is back under water
Activating Systems
When all spaces on a gauge are marked, the First Mate announces it to the Captain.
The Captain can then activate that system at any time.
The First Mate can activate only the drone and sonar systems, while the Captain can activate any system.
To activate a system, the Captain or First Mate does the following:
- Confirm with the Engineer that there are no breakdowns affecting the system. If there are, they must be repaired before the system can be activated.
- Announce "STOP" and raise a fist. All other players stop what they are doing and listen.
- Announce a system to activate. For example, "I'M DROPPING A MINE".
- Resolve the system activation (see below).
- Play resumes as normal.
Note: A system can be activated only if no symbol corresponding to the system has been crossed out.
Launching A Torpedo

The Captain can launch a torpedo at a space one to four spaces away from his submarine (not diagonally).
The Captain announces the point of impact (for example, "TORPEDO LAUNCHED, IMPACT IN C6"), but does not draw the trajectory on his sheet. Then the First mate erases all marks on the torpedo gauge.
A torpedo impact has the same outcome as a triggered mine (see Dropping a Mine, to the right). Then play resumes as normal. If a torpedo impacts in a space with a mine, the mine is destroyed; the Captain erases that mine from his sheet.
Note: A torpedo can damage the submarine that launched it!

Dropping A Mine

The Captain draws an M (for "mine") on a space adjacent to his submarine.
The Captain cannot drop a mine in a space on his route (with a line drawn in it), and a submarine cannot move into a space with its own mine.
The Captain announces "MINE DROPPED" and the First Mate erases all marks on the mine gauge. Then play resumes as normal.
At any time, except while surfaced, the Captain can trigger a mine by raising a fist, announcing TRIGGERING A MINE", and stating the mine's location. For example, "TRIGGERING A MINE AT G7".
There are three possible outcomes of a triggered mine or torpedo impact:
-
The enemy submarine is more than one space away from the mine or torpedo impact: The enemy Captain announces "ALL CLEAR" and play resumes as normal.
-
The enemy submarine is located in a space adjacent to the mine or torpedo impact:
The enemy Captain announces "INDIRECT HIT, ONE DAMAGE".
His First Mate crosses out the leftmost empty damage space on his sheet, then play resumes as normal.
-
The enemy submarine is located on the same space as the mine or torpedo impact:
The enemy Captain announces "DIRECT HIT, TWO DAMAGE".
His First Mate crosses out the two leftmost empty damage spaces on his sheet, then play resumes as normal.
Note: A mine can damage the submarine that dropped it!

Launching A Drone

Drones allow a team to narrow down the sectors in which the enemy's submarine could be located (the map is divided into nine sectors in real-time mode and four sectors in turn-by-turn mode). When a teammate launches a drone:
The teammate asks the enemy Captain if the enemy submarine is located in a particular sector. For example, "ARE YOU IN SECTOR 5?"
The enemy Captain answers "YES" or "NO" truthfully. The First Mate erases all marks on the drone gauge, then play resumes as normal.

Activating Sonar

When a teammate activates sonar, he announces "ACTIVATING SONAR". The enemy Captain announces two pieces of information about his submarine's location. This can include the row, column, or sector in which the submarine is located.
One piece of information must be true, the other must be false.
Each piece of information must be of a different type (row, column, or sector).
Then the First Mate erases all marks on the sonar gauge and play resumes as normal.

Activating Silence

When the Captain activates silence, he announces "ACTIVATING SILENCE" and moves his submarine without announcing the route.
He can move the submarine up to four spaces in a straight line without crossing an island or his own route.
The Captain indicates to his team the direction they are moving by gesturing with his hand behind the screen. The First Mate marks an empty space on a gauge of his choice and the Engineer crosses out a symbol on his sheet corresponding to the indicated direction. The First Mate erases all marks on the silence gauge, then play resumes as normal.

Activating The Scenario System

This system is not used in every scenario. It's function is determined by the scenario, which also indicates whether the gauge is considered to have four or six spaces.
End of the Game
When one submarine has suffered 4 damage, it is destroyed and the game ends.
The surviving team wins the game!
GENERAL ADVICE
The key to winning is good communication. If no one talks or no one listens, all hope is lost.
A good Radio Operator makes a big difference. Finding and tracking the enemy submarine is vital.
Continue Reading

